Overview
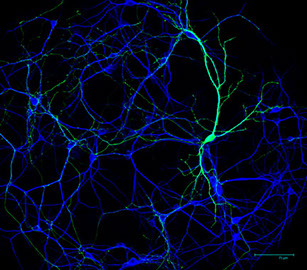
The brain contains billions of cells called neurons that wire together into an amazingly complicated circuit. During development, neurons form synapses, which are special neuron-to-neuron connections crucial for sending and receiving information. In theory, synapses can form anywhere one neuron contacts another neuron but, in actuality, synapses only form at a small percent of these sites. This feature generates specificity in neural circuits and is essential for proper brain function. Our experiments are aimed at understanding two main questions. 1: How do neurons recognize their correct synaptic partners during development? 2: How do neurons build the correct type of synapse? To accomplish these scientific goals, the lab uses a variety of molecular techniques including mouse genetics, viral vectors, biochemistry, electrophysiology, light and electron microscopy.
Synapses and Disease
It is now clear that many neurodevelopmental and psychiatric disorders are not caused by huge malformations of the brain but instead by subtle morphological or functional changes in specific types of synapses. Many genes involved in synapse development, including the cadherins and Kirrels, are linked to neurological disorders by genome-wide associational studies in humans but the cellular basis of how these genes may cause disease remain virtually unknown. A major goal of our lab research is understanding the cellular mechanisms by which disease-associated genes impact synaptic development using the mouse hippocampus as a model system.
Projects
Understanding Kirrel3 function in synapses and circuits
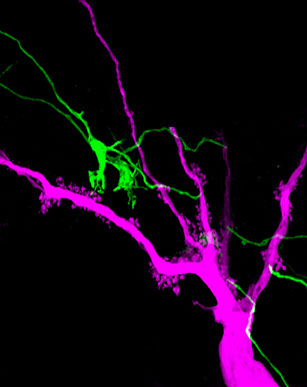
Kirrel3 is a cell adhesion molecule and mutations in the Kirrel3 gene are associated with autism and intellectual disabilities. Our lab is working to understand the function of Kirrel3 in the developing brain. We discovered that Kirrel3 is required for normal formation of mossy fiber filopodia, which are synaptic structures connecting excitatory DG neurons to GABAergic inhibitory neurons. These structures are important regulators of feed-forward inhibition in the hippocampus and consistent with this, the loss of Kirrel3 causes over-excitation of hippocampal neurons. We are now investigating the precise synaptic defects caused by Kirrel3 loss, elucidating Kirrel3 signaling mechanisms, and how Kirrel3-expressing neurons regulate hippocampal circuits and mouse behavior.
Cadherins in Synapse formation and function
We are testing the hypothesis that cell adhesion molecules make up a synaptic code in which different sets of adhesion molecules are present at different types of synapses to direct synapse specificity. The idea is not new but it remains stubbornly difficult to test experimentally. The classic cadherin family of cell adhesion molecules are well suited to play this role. We discovered that cadherin-9/cadherin-9 interactions are required at one type of hippocampal synapse and that cadherin-9/cadherin-10 interactions are needed at a different type of hippocampal synapse. Currently, we are developing new tools to help us determine the precise subcellular location of different cadherins, which may help explain mechanisms related to our past results and start to unravel the details of a synaptic cell adhesion code.
Neurobiology of suicide
In collaboration with Dr. Hilary Coon’s lab at Utah, we are working to identify cellular and synaptic changes associated with increased risk of suicide. This is an exciting new project to translate our expertise in model systems to a uniquely human brain disorder – look for updates!