Favorite Figures
Click figure for more information.

Click figure for more information.





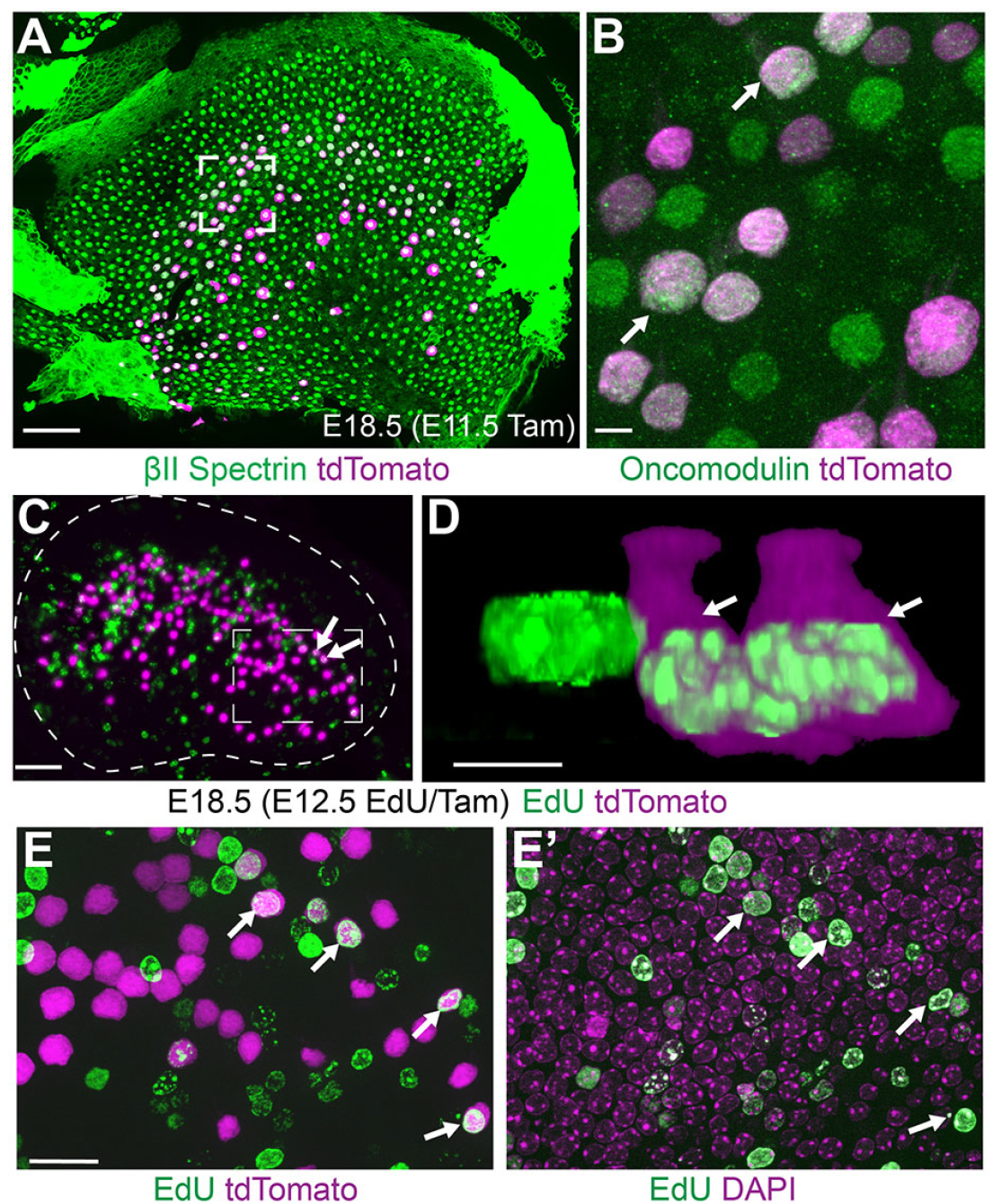
In this experiment Evan Ratzan demonstrated that the identity of Type I vestibular hair cells is determined shortly after mitosis in the developing mouse utricle. He did this by simultaneously labeling mitotic cells with EdU while genetically labeling the differentiating Type I hair cells in FGF8 mcm transgenic mice (panels C-E). This was remarkable because prior to this finding, it was thought that vestibular hair cell types could only be distinguished from each other after birth.
See Ratzan, Moon, Deans, Development, 2020

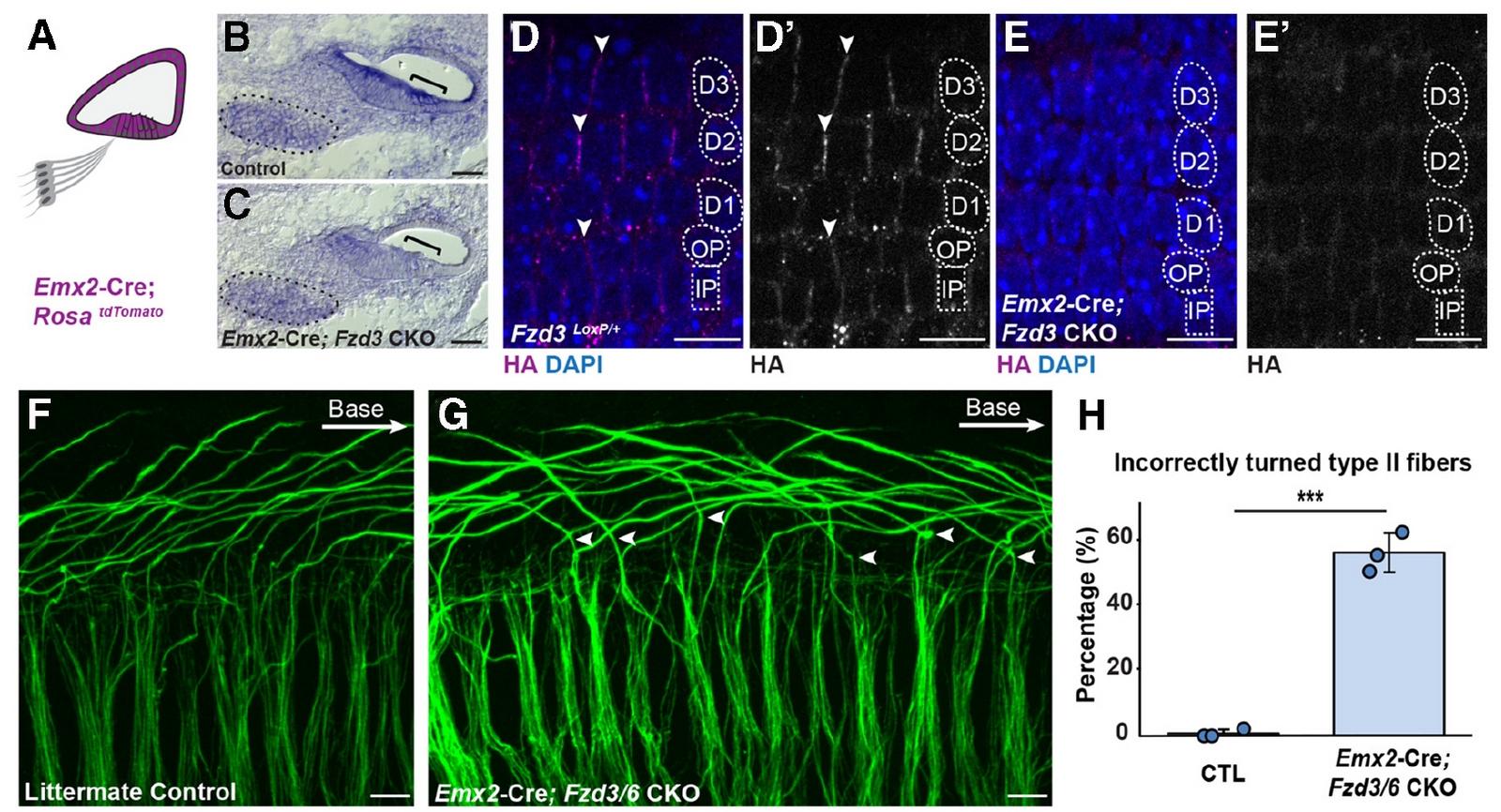
Using a detailed series of conditional knockout mice, Satish Ghimire demonstrated that the Core PCP proteins function non-cell autonomously from supporting cells of the cochlear duct to guide axon turning. In this example, removing Frizzled3 and Frizzled6 from the organ of Corti results in the random turning of Type II Spiral Ganglion Neuron peripheral axons (panels F-H). This is significant because during axon guidance in other systems, the PCP proteins function within the growth cone itself.
SeeGhimire and Deans, J. Neuroscience, 2019 and Ghimire, Ratzan, and Deans, Development, 2018

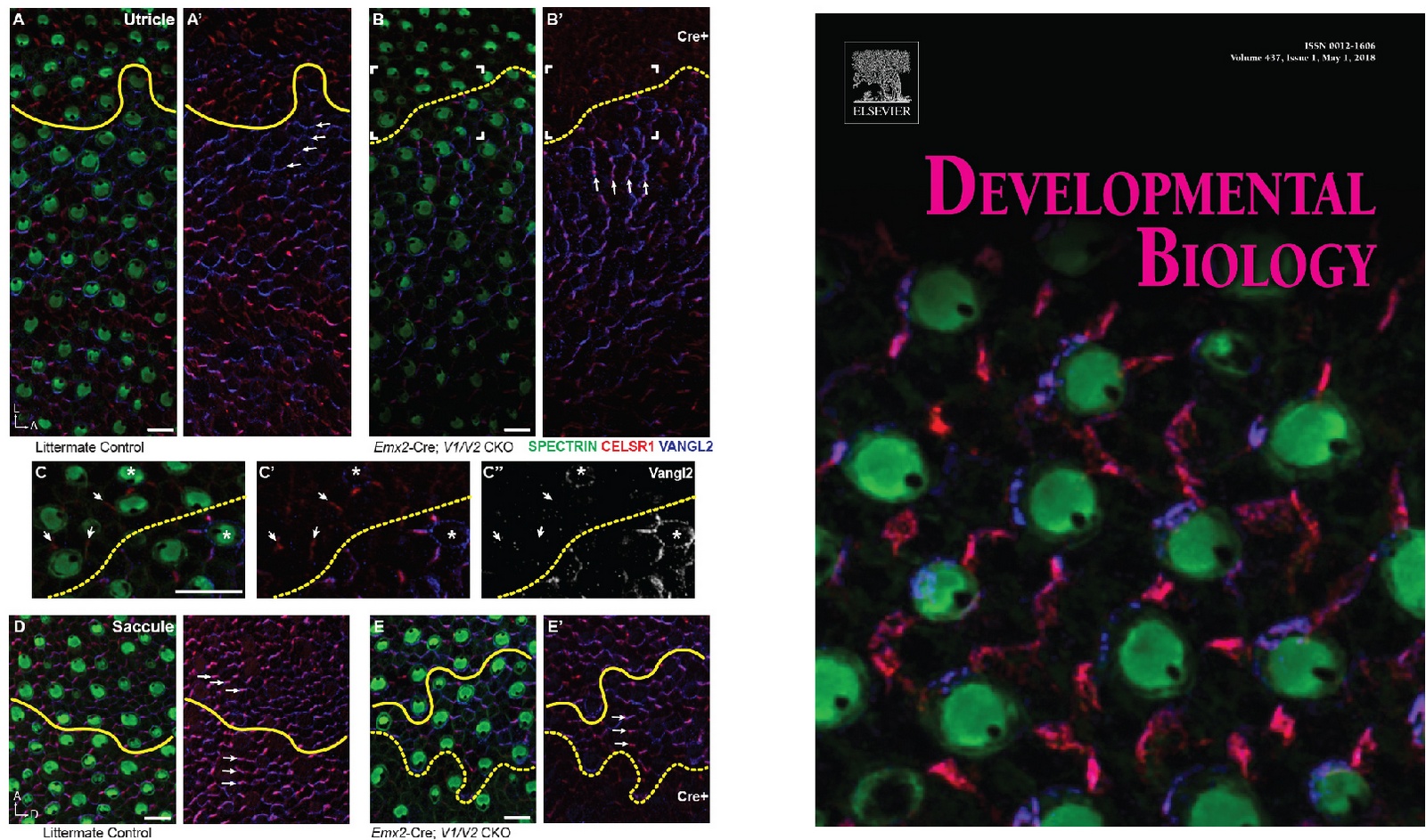
Michelle Stoller was able to show domineering non-autonomy using Cre recombinase to generate ‘clonal’ boundaries between wild type and mutants cells in the mouse utricle (panels A,B). This was significant because it demonstrated that the supporting cells contributed to PCP signaling and coordinated the orientation of neighboring hair cells. Not only was this our favorite figure from her postdoc, it provided inspiration for the journal cover.
See Stoller et al., Developmental Biology, 2018

Using an image captured by Satish Ghimire we provided an
exciting cover for Developmental Biology. Our review in this edition highlights
Satish’s dissertation work and his contributions to our understanding of
cochlear innervation.
See Deans MR. Dev Biol. March 2022

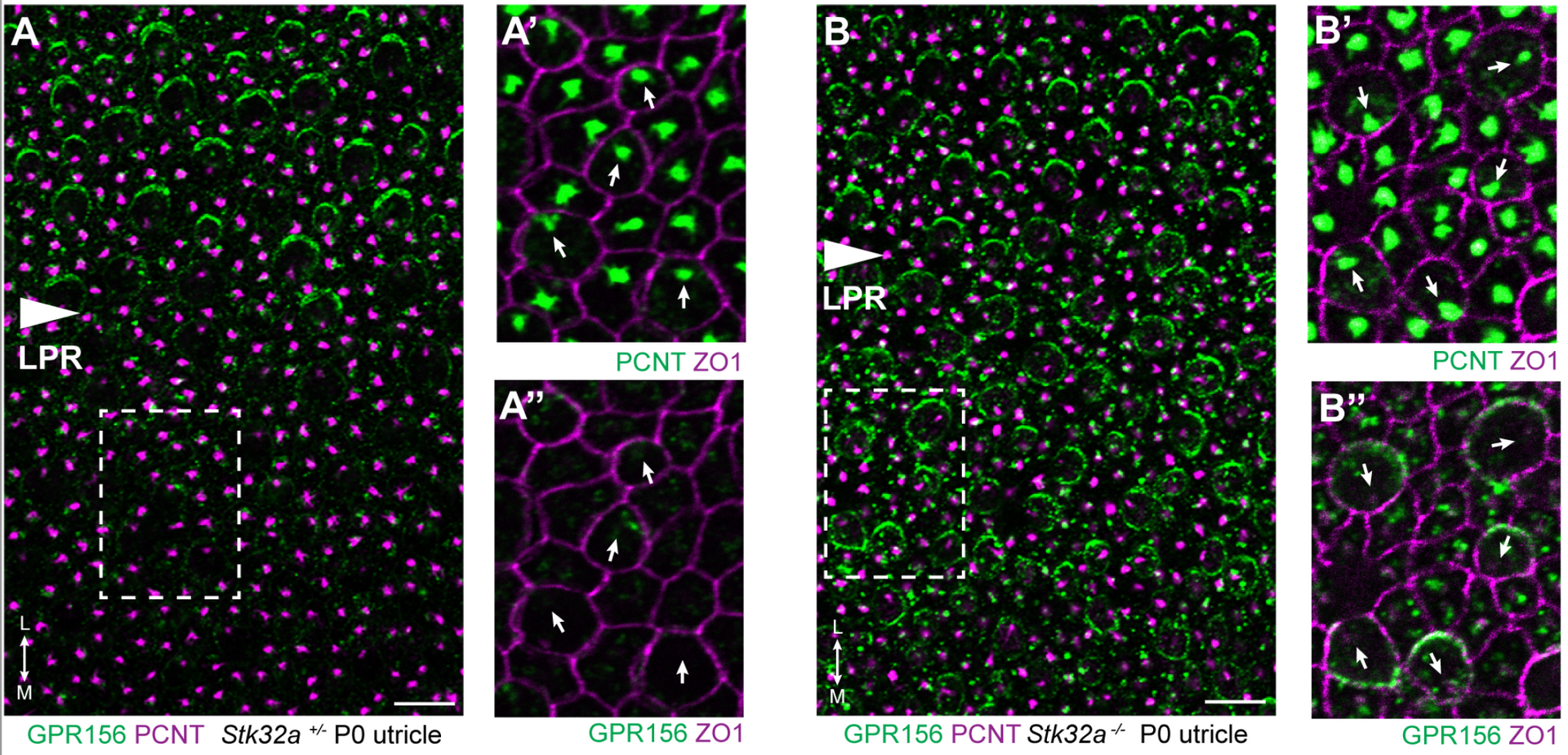
A long effort initiated by Evan Ratzan culminated in this exciting collaboration with Basile Tarchini from Jackson Labs. Together we demonstrated that STK32A (a kinase with no known prior function) acts within the inner ear to regulate GPR156 distribution in hair cells and thereby position the Line of Polarity Reversal in the developing utricle and saccule. See Jia and Ratzan et al. 2023